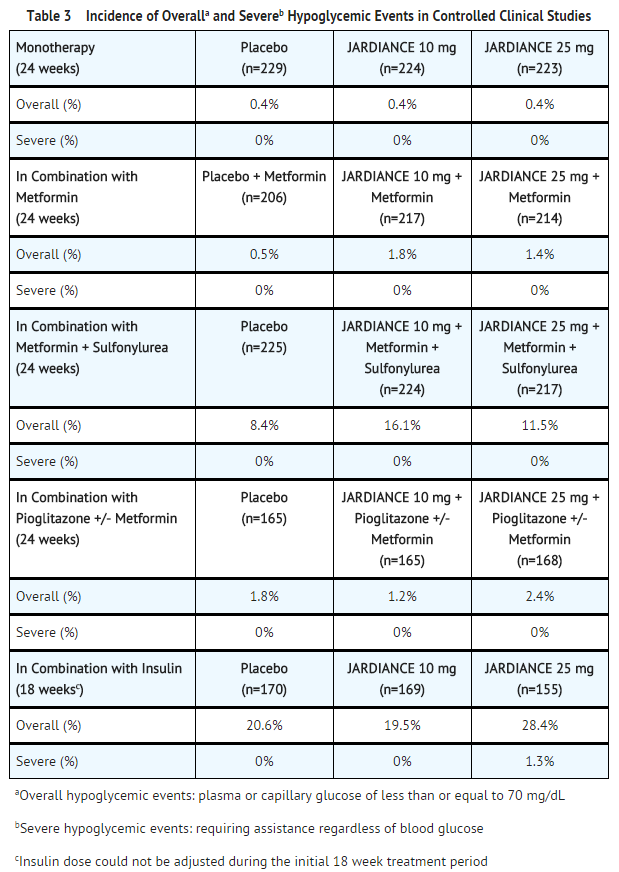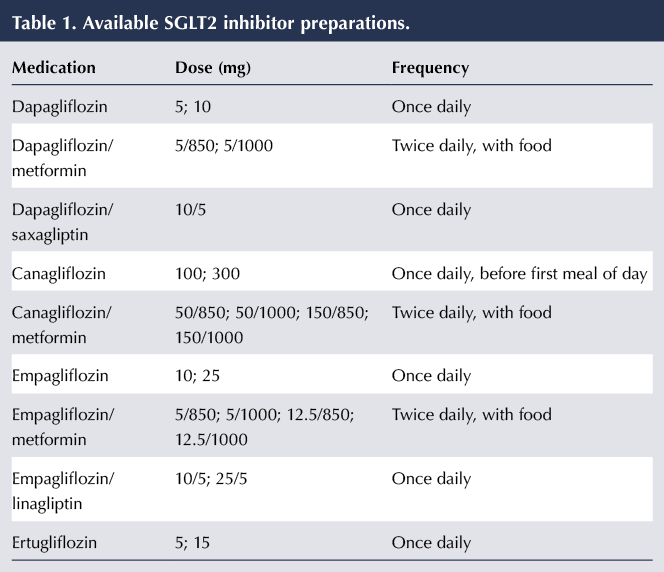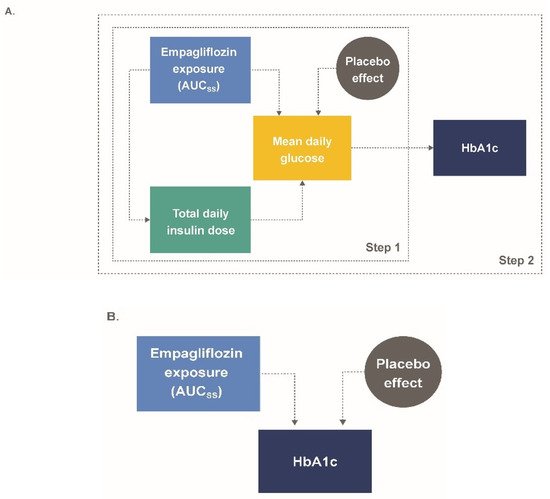
Pharmaceutics | Free Full-Text | A Model-Informed Drug Development (MIDD) Approach for a Low Dose of Empagliflozin in Patients with Type 1 Diabetes
U.S. FDA approves first-in-class Glyxambi® (empagliflozin/linagliptin) tablets for adults with type 2 diabetes

Fixed dose combinations of empagliflozin/linagliptin for 52 weeks as add-on to metformin in subjects with type 2 diabetes - Media Centre | EASD


![Farxiga vs. Jardiance [Diabetes Drug Comparison] Farxiga vs. Jardiance [Diabetes Drug Comparison]](https://www.drugwatch.com/wp-content/uploads/jardiance-overview-640x0-c-default.png)